pH describes amount of H+ cations in solution. Concentration of these ions can change in very wide range - most often it has values lying somewhere between 1M and 10-14M, although sometimes even higher and lower concentrations can be observed.
Using numbers differing by many orders of magnitude is impractical, thus in 1909 Danish biochemist Søren Sørensen developed the pH scale and introduced pH definition - minus logarithm base 10 of [H+]:
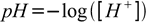
While pH defined this way can be usefull when doing simple pH calculation for diluted solutions of acids and bases, it is not correct in general. Behavior of the ions in the solution depends not on their concentrations, but on activities. In case of very diluted solutions (say below 0.001M) difference between concentration and activity can be neglected. In case of more concentrated solutions - it have to be taken into account. Thus in reality more precise definition is used:
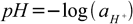
and is sometimes called 'thermodynamic pH' as opposed to 'concentration pH'. What is important is the fact, that all pH meters measure thermodynamic pH values, as present in the Nernst equation.
For more information see pH definition lecture (and other lectures on pH) at ChemBuddy - chemical calculators site.


